

Section A: What are IPEDs and how do they fit within the field of Human Enhancement Drugs?
Human enhancement drugs (HEDs) are substances taken with the intention of positively (from the user’s perspective) manipulating one or more aspects of appearance and/or physical or cognitive function (McVeigh et al, 2012, Evans-Brown et al, 2012). HEDs can be categorised into six main groups, as shown in Table 1 (McVeigh et al, 2012).
Table 1: Categories of Human Enhancement Drugs
| Drug Category | Examples of Specific Drugs |
|---|---|
| 1. Musculature | Anabolic steroids, Growth Hormone |
| 2. Weight Loss | Rimonabant, Sibutramine, 2,4-dinitrophenol (DNP) |
| 3. Skin & Hair Appearance | Creams containing mercury, Melanotan, Latisse |
| 4. Sexual Function | Sildenafil, Bremelanotide, Yohimbine |
| 5. Cognitive Function | Methylphenidate, Modafinil, Piracetam |
| 6. Mood and Social Behaviour | Fluoxetine, Beta-blockers, Diazepam |
Image and performance enhancing drugs (IPEDs) form a sub-group of HEDs, principally associated with changes in physical performance or appearance and/or weight loss. The demarcation between IPEDs and the broader family of HEDs is best described via the motivations behind self-directed use. Whilst specific drugs may be used for more than one purpose (e.g. melanotan; primarily used for skin-tanning but may also be used to enhance sexual function), the primary motivation for their use can define them as IPEDs. Drugs in the first two categories (musculature and weight loss) can therefore be defined as IPEDs, along with melanotan. Drugs within the remaining categories and other pharmaceutical drugs may be used alongside IPEDs to alleviate or prevent side-effects of IPED use. Within the broader literature on IPED use, the focus is primarily on those who use anabolic-androgenic steroids (AAS) and the range of drugs that provide additional image or performance enhancement (e.g. Growth hormone, insulin) or help to address common side-effects of AAS use.
The use of substances to enhance some aspect of an individual’s physical or emotional state is not a new phenomenon. References to the use of such substances can be found throughout human history and many of the more popular drugs being used today have been in self-directed use for decades or longer (Evans-Brown et al, 2012). In particular, the use of anabolic steroids for performance enhancement can be traced back to the consumption of bull testes in Ancient Rome and subsequently to the work of Brown-Sequard in the late 1800s, producing his own ‘elixir’ from animal testes, marketed as a means to ‘restore vitality’ (Matfin, 2010). Later work by Lacquer in 1935, isolated testosterone for the first time and paved the way for the proliferation of anabolic steroids we see today (Nieschlag & Nieschlag 2014). Whilst originally developed for a range of therapeutic purposes, the off-label use of anabolic androgenic steroids (AAS) in bodybuilding and weightlifting was noted in the late 1940’s and subsequently diffused to competitive sports and the wider population (Evans-Brown et al 2012). The marketing of other drugs for issues such as weight loss, changes in skin appearance, anxiety, sexual dysfunction and a range of others issues, has an equally long and troubled history (Evans-Brown et al 2012) prompting the British Medical Association (BMA) to release two books in the early 1900s, detailing the market in these unlicensed drugs and raising awareness of the many potential health issues (BMA 1909, 1912). The marketing of these drugs mirrors that seen today, however the range of drugs has since grown, as has means to disseminate such marketing to a wider audience via the internet. The proliferation of online sources for IPEDs and illicit or counterfeit medicines presents a unique challenge for authorities attempting to control or prevent distribution, with an estimated global industry worth in excess of $75 billion per year (Di Nicola et al, 2015). In particular, there is emerging evidence of a wide spectrum of fake and illegal online pharmacies, many of which sell poor quality products and/or act as phishing sites, but there exists a small number of sites that appear to take a more customer focused approach, attempting to maintain high levels of customer satisfaction (van de Ven & Koenraadt, 2017; Di Nicola et al, 2015). It seems likely that this market will continue to grow and as such, there is an increasing need for research and practice to evolve alongside it in order to meet the emerging challenges it presents.
References: Section A
British Medical Association (1909) Secret remedies. What they cost and what they contain. British Medical Association
British Medical Association (1912) More secret remedies. What they cost and what they contain. British Medical Association
Di Nicola, A., Martini, E., Baratto, G., Antonopoulos, G. A., Boriero, D., Da Col, W., et al. (2015). FAKECARE: Developing expertise against the online trade of fake medicines by producing and disseminating knowledge, counterstrategies and tools across the EU. eCrime Research Reports No.2. Trento: eCrime — University of Trento.
Evans-Brown, M., McVeigh, J., Perkins, C., & Bellis, M. A. (2012) Human enhancement drugs: The emerging challenges to public health. Liverpool: Public Health Observatories in England
Matfin, G. (2010) The rejuvenation of testosterone: philosopher’s stone or Brown-Sequard Elixir? Ther Adv Endocrinol Metab 1(4) 151:154 DOI: 10.1177/2042018810385052
McVeigh, J., Evans-Brown, M. & Bellis, M.A. (2012) Human enhancement drugs and the pursuit of perfection ADICCIONES 24:3 • pp:185-190
Nieschlag, E. & Nieschlag, S. (2014) Testosterone deficiency: a historical perspective Asian Journal of Andrology 16, 161–168; doi: 10.4103/1008-682X.122358
van de Ven K, & Koenraadt R. (2017) Exploring the relationship between online buyers and sellers of image and performance enhancing drugs (IPEDs): Quality issues, trust and self-regulation. Int J Drug Policy. Dec;50:48-55. doi: 10.1016/j.drugpo.2017.09.004. PMID: 29028563.

There is a vast and varied list of substances that can be categorised as IPEDs and a comprehensive examination of all of them is beyond the scope of this site. The focus here will be on the key drugs frequently employed by most IPED users, broken down by category.
- Musculature
- Weight Loss
- Skin Appearance
- Ancillary Drugs (For Side Effects)
Section B: Musculature
Anabolic-androgenic steroids (AAS)
The most commonly used drugs amongst IPED users are, without question, the anabolic steroids (Begley et al, 2017). Synthetic derivatives of the primary male sex hormone testosterone (and related hormones), anabolic steroids were originally pharmaceutical preparations, developed to treat a range of medical conditions (e.g. breast cancer, anaemia, recovery from injury or hormone replacement therapy, amongst others). All synthetic anabolic steroids are developed from a base of one of three parent steroids (testosterone, dihydrotestosterone & nandrolone), all of which occur naturally in the human body.
The properties of a particular anabolic steroid, including the effects and side-effects, are largely determined by the parent steroid and subsequent manipulations of the molecular structure (Kicman, 2008). These manipulations are designed to take advantage of specific properties of each parent steroid, whilst attempting to minimise the generally less desirable oestrogenic and androgenic side-effects. Oral steroids have a further manipulation that enables the steroid to survive first pass metabolism by the liver, ensuring that more of the active ingredient is bio-available (Kicman, 2008). However, this additional manipulation also presents increased risk of hepatotoxicity.
All anabolic steroids can be defined in terms of their anabolic/androgenic ratio, where anabolic refers to the primary, desirable, characteristics (e.g. muscle building, recovery) and androgenic refers to the secondary effects, that may or may not be desirable (growth of body hair, deepening of voice, increase in libido). The androgenic properties of testosterones can be a particular issue in female IPED users. As a general rule, the more androgenic an anabolic steroid is, or the more easily it converts to oestrogen (a process called ‘aromatisation’), the greater the likelihood of adverse side-effects. For example; dihydrotestosterone (DHT) does not aromatise and therefore derivatives of this steroid (for further information see https://www.ipedinfo.co.uk/ipeds-terminology-explained/ e.g. stanozolol, mesterolone) do not carry significant risk of oestrogenic side-effects such as gynaecomastia, however anabolic steroids derived from DHT are typically weaker than nandrolones or testosterones in terms of their anabolic properties (Kicman, 2008). It is these variations in the properties of individual anabolic steroids that frequently leads users to employ multiple steroids in a cycle, in order to take advantage of differing effects, often in the belief that those differing effects may work synergistically. This frequently includes both oral and injectable anabolic steroids being used simultaneously (Begley et al, 2017). It is important to note that combining different steroids not only potentially combines their primary effects, but also their side-effects. Injectable anabolic steroids are typically injected intramuscularly, ideally in larger muscles such as the quadriceps or gluteus, however some users may also inject smaller muscles such as the deltoid, which can, in some cases, result in adverse reactions at the injection site.
It is important to note that, as with all IPEDs, the lack of properly regulated, reliable sources to purchase these drugs, results in a high proportion of poor quality ‘street-level’ products. They may contain an anabolic steroid but it may not be the actual steroid, or dose, stated on the label. There may also be other drugs or a variety of contaminants or adulterants within the product (Kimergard et al, 2014) Consequently, it is important to note that user-reported experiences of using particular anabolic steroids (or indeed, any IPED) may not relate to known effects or side-effects for the drug they believe they used. Equally; they may find unexpected side-effects as a result of these issues around quality control.
Table 2: Examples of commonly used anabolic steroids (by parent steroid):
Parent steroid
Common AAS
Example images
Parent steroid
Testosterone:
Most potent in terms of muscle building, but also most prone to aromatisation and androgenic side-effects
Common AAS
Testosterone Esters: Injected
Esters are chemical additions to the parent molecule to regulate rate of absorption. The longer the ester, the slower the rate at which serum testosterone levels increase and the less frequently injections are required. Examples of esters (in increasing length): Acetate, Propionate, Enanthate, Cypionate, Phenylpropionate, Decanoate.
Example images

Parent steroid
Testosterone
Common AAS
Boldenone (aka Equipoise): Injected
A veterinary steroid with strong anabolic and moderate androgenic properties. Both oestrogenic and androgenic side-effects are possible with this steroid.
Example images

Parent steroid
Testosterone
Common AAS
Sustanon: Injected
This is a blend of four different esterified testosterones; designed to allow a rapid rise in testosterone levels, followed by slower release testosterones that maintain elevated serum levels over (approximately) 3 weeks.
Example images

Parent steroid
Testosterone
Common AAS
Methandrostenolone (aka Dianabol): Oral
Like all oral AAS, methandrostenolone is a methylated compound (chemically altered to survive passing through the liver) and as such, has the potential to be hepatotoxic. The use of methandrostenolone can produce significant water and fat gain and carries the potential to induce gynaecomastia.
Example images

Parent steroid
Testosterone
Common AAS
Fluoxymesterone (aka Halotestin): Oral
Fluoxymesterone is an oral AAS derived from methyltestosterone. Unlike the parent steroid, fluoxymesterone does not offer significant anabolic properties, but can be used for strength gains with minimal muscle gain. It does not aromatise and should therefore not present oestrogen-related side-effects. It is methylated and can therefore, adversely impact liver function alongside the other side-effects common to all AAS.
Example images

Parent steroid
Dihydrotestosterone (DHT):
DHT derived AAS are characterised by lean muscle gains, largely due to the lack of androgenic side-effects including fat and water gain. They are also less likely to produce oestrogenic side-effect as DHT derived AAS cannot be aromatised into oestrogen.
Common AAS
Drostanolone (aka Masteron): Injected
As with other DHT-derived AAS, Drostanolone is not generally used to build significant mass, but rather to build lean muscle or maintain muscle whilst dieting to lose fat.
Example images

Parent steroid
Dihydrotestosterone
Common AAS
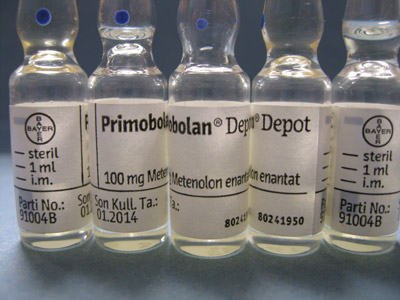
Methenolone (aka Primobolan): Injected or Oral
Primobolan is a widely used AAS and, as with most DHT derived steroids, provides lean muscle gain. Available in two different preparations; an oral version using the acetate ester and an injectable using the longer enanthate ester. The oral version is not methylated like other oral AAS and as such, is less likely to adversely impact the liver.
Example images
Parent steroid
Dihydrotestosterone
Common AAS
Stanozolol (aka Winstrol): Injected or Oral
Unlike other AAS, Stanozolol can be administered via injection or orally, the compound is the same in both cases. The injectable version is a water-based solution, rather than oil-based, (as all other injectable AAS are) and therefore requires frequent, possibly daily, injections.
Example images

Parent steroid
Dihydrotestosterone
Common AAS
Mesterolone (Proviron): Oral
Although mesterolone is a weak anabolic agent, it is often used alongside other AAS as it is believed to have a synergistic effect with them. It may also be used as an anti-oestrogen or to increase libido.
Example images

Parent steroid
Dihydrotestosterone
Common AAS
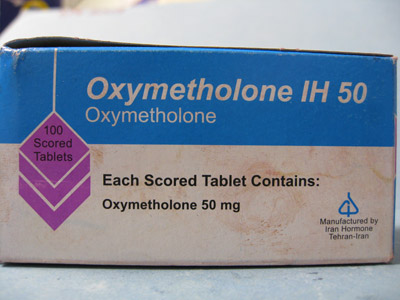
Oxymetholone (aka Anapolan or Nap-50): Oral
Characterised by rapid gains in weight, strength and muscle mass, oxymetholone is a popular oral steroid. Unlike other DHT derived AAS however, oxymetholone can produce significant water retention and carries significant risk of oestrogenic side-effects.
Example images
Parent steroid
Dihydrotestosterone
Common AAS
Oxandrolone (aka Anavar): Oral
Oxandrolone is a popular steroid for those wishing to retain muscle when dieting or those seeking strength gains without significant muscle mass. Oxandrolone does not produce significant oestrogenic or androgenic side-effects but as with all oral AAS carries some risk of hepatotoxicity.
Example images

Parent steroid
Nandrolone (aka 19-Nor Testosterone)
Nandrolone derived AAS exert powerful anabolic effects but with much less androgenic activity (and therefore androgenic side-effects) than testosterones (Kicman, 2008). Nandrolones also aromatise more slowly than testosterones and therefore have reduced risk of oestrogenic side-effects, although they are still possible at high doses.
Common AAS
Nandrolone decanoate (aka Deca Durabolin or just ‘Deca’): Injected
Widely used by bodybuilders, nandrolone decanoate can produce significant lean strength and muscle gains with relatively low androgenic or oestrogenic side-effects compared to testosterone. However, high doses and/or long-term administration can produce these effects.
Example images

Parent steroid
Nandrolone
Common AAS
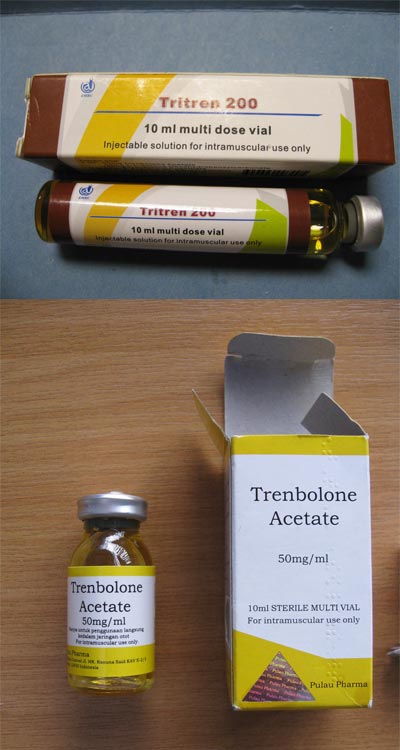
Trenbolone: Injected
Popular and powerful anabolic agent with a reduced possibility for aromatisation as compared to both the parent nandrolone and testosterone. However, trenbolone does exert more androgenic activity than nandrolone. Trenbolone can be esterified and is typically found with either the short acetate ester or the much longer enanthate ester. There is also a version (called Parabolan) using the hexahydrobenzylcarbonate ester. This last version is the only one that has ever been licenced for human use, although its use is now discontinued. Other preparations of trenbolone are only licenced as veterinary steroids. Anecdotal reports suggest trenbolone carries significant potential for psychological side-effects (irritability, anxiety, depression, paranoia have all been reported).
It is possible to find an oral preparation known as methyltrienolone, although its use appears rare. Technically this is a derivative of trenbolone, rather than simply an oral version. It exhibits potent anabolic and androgenic effects and carries significant risk of hepatotoxicity.
Example images
Anabolic Steroid Blends
 As noted in Table 1, the commonly used AAS sustanon is a blend of four different esterified testosterones, designed to provide a rapid rise in serum testosterone levels followed by a sustained elevation over several weeks due to the combination of fast, medium and slow release esters used. From a therapeutic perspective, this allows a reduced injection schedule for people undergoing hormone replacement therapy whilst maintaining relatively stable serum testosterone levels. Many underground laboratories (UGLs) have also adopted this approach; producing their own anabolic steroid blends of testosterones as well as blended trenbolones and nandrolones. Typically, such preparations are named to reflect the blend (i.e. TriTren) but not always (i.e. Test 400 is a blend of testosterones). Although many of the preparations use the same basic steroid (testosterone, trenbolone or nandrolone) but with varying esters, there are user reports emerging of blends that incorporate different steroids. From the user’s perspective, this may simplify the design of a cycle and reduce the frequency of injections required. However, it is important to note that some preparations appear ill-conceived with regard to the esters used and may therefore reflect a lack of understanding of steroid chemistry on the part of the manufacturer.
As noted in Table 1, the commonly used AAS sustanon is a blend of four different esterified testosterones, designed to provide a rapid rise in serum testosterone levels followed by a sustained elevation over several weeks due to the combination of fast, medium and slow release esters used. From a therapeutic perspective, this allows a reduced injection schedule for people undergoing hormone replacement therapy whilst maintaining relatively stable serum testosterone levels. Many underground laboratories (UGLs) have also adopted this approach; producing their own anabolic steroid blends of testosterones as well as blended trenbolones and nandrolones. Typically, such preparations are named to reflect the blend (i.e. TriTren) but not always (i.e. Test 400 is a blend of testosterones). Although many of the preparations use the same basic steroid (testosterone, trenbolone or nandrolone) but with varying esters, there are user reports emerging of blends that incorporate different steroids. From the user’s perspective, this may simplify the design of a cycle and reduce the frequency of injections required. However, it is important to note that some preparations appear ill-conceived with regard to the esters used and may therefore reflect a lack of understanding of steroid chemistry on the part of the manufacturer.
Peptides
Frequently used alongside AAS (Begley et al, 2017), peptides are injectable drugs that mimic naturally occurring hormones. Peptides are synthetically created strings of amino acids in differing combinations, with variations in the combinations producing different effects in the body. Peptides can usually only be injected (typically subcutaneously), there are no viable, orally active preparations available. The one exception to this is a nasal spray delivery system for Melanotan. Whilst it appears to be less effective than injectable preparations, it has grown in popularity since its inception (Evans-Brown et al, 2012). All peptides, with the exception of insulin, are sold as freeze-dried powders that require reconstitution with bacteriostatic water before administration. Care must be taken both when reconstituting the peptide and storing it (ideally in a fridge); because the peptides are fragile and can be damaged by improper handling, and because the additional preparation step provides an additional opportunity for bacterial contamination (Sigma Aldrich handling guide).
Whilst they can be used for their anabolic properties (e.g. Human Growth Hormone (HGH), Insulin, IGF-1) peptides have a more diverse range of primary effects than AAS. Human growth hormone for instance, can be also be used for its potentially positive effects in fat loss and in fact there exists a derivative of HGH known as Human Growth Hormone Fragment 176-191, which is specifically designed to make use of the ‘fat burning’ properties of HGH. Melanotan, whilst primarily used for its skin darkening properties, is sometimes also used for its perceived positive effects on libido (Evans-Brown et al 2012). This diversity of effect may also influence the user’s decision whether to include peptides in their cycle.
For those using peptides for their anabolic properties, the most commonly used appears to be human growth hormone. Whilst prevalence of its self-directed use is difficult to calculate accurately, there is evidence of significantly increased use amongst IPED users in the UK (Evans-Brown & McVeigh, 2009a, Begley et al, 2017). Other anabolic peptides appear to be less used but still appear frequently in self-reported patterns of use (e.g. Insulin, IGF-1, GHRP, MGF).
SARMs
Selective Androgen Receptor Modulators (SARMs) are relatively new additions to the family of IPEDs. Early work on developing these drugs started in the late 1990’s but potentially viable preparations have only been available since 2015 or later (Solomon et al, 2019). SARMs are non-steroidal drugs, taken orally, that seek to mimic the anabolic action of anabolic steroids, whilst avoiding the androgenic or oestrogenic side-effects. As SARMs do not aromatise they have no oestrogenic effects and current evidence suggests minimal androgenic effects. However, it is important to note that at this time (2020) very few SARMs have undergone clinical trials. Of the five seemingly most popular, or widely available, SARMs, only two (Ligandrol/LGD-4033 & Enobosarm/Ostarine) have completed phase 1 clinical trials and only Enobosarm has been extensively studied in human trials. Consequently, information on effects and side-effects of these drugs is extremely limited, although there are some indications that they can increase muscle mass and bone density, as well as other, potentially therapeutic, effects (Solomon et al, 2019).
From a clinical perspective, if SARMs can be developed that present an acceptable safety profile and fewer side-effects than anabolic steroids or related drugs; then they offer a range of potential benefits in treating conditions previously treated with AAS. Currently, SARMs are being developed to treat conditions such as osteoporosis, benign prostatic hyperplasia (BPH), muscular dystrophy and cachexia, amongst others (Solomon et al, 2019). Enobosarm is currently the most promising of these drugs but, whilst there are a number of clinical trials ongoing, it has not yet (as of 2020) been licenced as a medicine anywhere globally.
Despite the relative lack of clinical evidence base, or formal approval by government health bodies, SARMs are widely and freely available from internet sources globally and as with all IPEDs, there exists a large user community providing advice around their use. SARMs may be used alongside traditional anabolic steroids or as an alternative. They are also sometimes used in order to ‘bridge’ between steroid cycles due to their perceived comparatively minor side-effects. It should be noted that many websites selling SARMs also sell a number of other drugs often described as SARMs, either in marketing or in user forums, but they are not actually SARMs. That is, they do not share the same chemical structure as SARMs. Notable amongst these are cardarine and ibutamoren (MK-677). Whilst they are not strictly speaking SARMs, they will be discussed here as they are generally grouped with SARMs by users and distributors and they are not structurally related to anabolic steroids or peptides, although ibutamoren is somewhat similar to other growth hormone promoting drugs. A search of google trends shows a rapid increase in searches for SARMs worldwide, from 2014 to 2020, with a slight drop around April and May 2020, possibly due to the influence of lockdown restrictions as a result of Covid-19. Enobosarm appears to be the most searched for SARM (Google trends).
Commonly used SARMs (and related drugs)
All of the drugs listed here are taken orally and all are sold as research chemicals only. Typical doses stated here are for male users, female users would typically take approximately half the stated male dose (Llewellyn 2017). There are no licenced preparations of any of these drugs currently (2020) available globally, except as research chemicals. No licence is needed to purchase them.
Cardarine (aka: GW-501516):
Cardarine is a PPARD (Peroxisome Proliferator-Activated Receptor Delta) receptor agonist, usually sold (and often used) alongside SARMs. Early studies, including Phase I & II human trials, showed significant improvement in serum lipid profiles; improving HDL vs LDL ratios and showing some signs of lean muscle gain (Olson et al, 2012). Typically taken in doses of around 10mg per day, for 6-12 weeks. Despite apparently completing early human trials successfully, the development of this drug ceased in 2009 following the discovery of extensive cancer development in all animal study subjects, at all doses. There is therefore likely to be no further development of this drug by pharmaceutical companies.
Ibutamoren (aka: MK-677):
Ibutamoren is an orally active drug that mimics the action of Growth Hormone Releasing Peptides, in particular it is functionally identical to GHRP-6 (Murphy et al, 1998), increasing natural levels of human growth hormone and IGF-1. Whilst an injectable version of this drug is available, it is commonly available and most widely used, as an oral drug with dosages of 5-25mg per day. Although the very limited trials thus far have noted no significant adverse side-effects, ibutamoren remains a research chemical with almost no human studies and no full clinical trials at this time (2020).
Andarine (aka: S4):
Originally developed as a potential treatment for BPH and cachexia, andarine is a widely used and popular SARM. It is believed to exhibit a relatively mild anabolic effect with relatively minimal side-effects. However, no extensive human clinical trials have been completed for this drug and as such, much of the data on side-effects comes from anecdotal reports among the IPED community. Notable amongst these are user reports of visual disturbance such as night blindness or a ‘yellow tint’ to day vision (Llewellyn, 2017). This does not appear to be a permanent side-effect and should resolve after use of the drug ceases. It appears unlikely that this drug will continue to be developed as the pharmaceutical company developing it is now more focused on Enobosarm. It is typically used at a dosage of 25-75mg per day for 6-12 weeks.
Enobosarm (aka: Ostarine, MK-2866)
Enobosarm is, like andarine, functionally similar to anabolic steroids but appears to offer a better safety profile. It has been extensively studied in a number of Phase II and III clinical trials as a potential treatment for a number of conditions, notably cachexia (muscle wasting) and appears to be well tolerated (Dalton et al, 2011). Despite this apparent success, development for treating cachexia does not appear to be continuing but the drug continues to be studied for a range of other conditions such as breast cancer. Enobosarm appears to have reasonable anabolic effects but is well tolerated, with relatively minor side-effects. Most notable amongst the side-effects is its adverse effects on the HDL/LDL ratio, with suppression of HDL in some patients. Whether this presents a clinically significant risk is unknown at this time. Enobosarm is typically taken for 6-12 weeks at a daily dose between 10-30mg and would appear to be the most popular SARM.
RAD-140 (aka: Testolone):
This drug is still in early pre-clinical trials but appears to show a potent anabolic effect with minimal androgenic effects. It has been trialed (in animal studies and in vitro) as a possible replacement for testosterone in hormone replacement therapy, shown distinct promise in treating breast cancer (Yu et al, 2017), and cachexia. There is also some evidence to suggest RAD-140 offers neuroprotective qualities that may make it a suitable treatment for alzheimer’s disease, although it must be noted this is only in animal studies at this stage. There is some suggestion it may be hepatotoxic, but this is based on a single case study (Flores et al, 2020). Anecdotal reports suggest the drug is well tolerated (Llewellyn, 2017) but the absence of human trials means reliable safety data do not currently exist. Typical dosing is 10-20mg per day for 6-12 weeks.
YK-11:
Unlike other SARMs, YK-11 is synthesised from the same molecular structure as testosterone, although it does not resemble any available anabolic steroid (Kanno et al, 2011). Whilst early studies suggest it may have potent anabolic properties and some androgenic effects (although potentially less than anabolic steroids) all research at this stage has been conducted in vitro only and as such, reliable data on effects and side-effects does not exist. Dosage is typically 5-10mg per day for 4-8 weeks. Anecdotal reports suggest it is a potent anabolic but even amongst the IPED community, it appears there is some confusion over-dosing schedules and effects (Llewellyn, 2017).
Ligandrol (aka: LGD-4033):
Ligandrol is the only other SARM (besides Enobosarm) to have undergone Phase I and II clinical trials. As with Enobosarm, significant increases in lean body mass were noted and the drug appears well tolerated. However, results showed suppression of natural testosterone and some adverse impact on cholesterol levels as with anabolic steroids. All effects were dose-related and suppression of free testosterone was demonstrated even at a dose of 1mg daily. These effects appeared to resolve over time when use of the drug was discontinued (Basaria et al, 2013). The same study showed a cumulative effect of dosing, with serum levels significantly higher at day 21 compared to day 1. It should be noted that, despite being well tolerated, these studies are very small-scale and short duration and with a maximum 1mg dose. Longer trials, with larger sample groups are required to establish more reliable long-term effects and side-effects. The suppression of natural testosterone noted is of particular concern, suggesting the need for post-cycle therapy similar to anabolic steroids. It may offer a viable alternative to injectable anabolic steroids, thereby avoiding injection related harms, if it can be shown to offer a better safety profile than traditional oral steroids, especially with regard to hepatotoxicity. That data are not yet available however. Ligandrol is typically used at a dose of 2-10mg per day for 4-8 weeks. Given the suppression noted at just 1mg per day, it seems likely it will be more severe at these doses.
Section B: Weight Loss
Weight loss products generally, are amongst the most widely used dietary supplements globally, with an extensive and diverse market from readily available over-the-counter products to the so-called ‘hardcore’ products featured here. Dietary products and supplements advertised as aiding (or directly causing) weight loss represent a ubiquitous multi-billion dollar global industry (Evans-Brown et al, 2012). Whilst the majority of the weight loss supplements commercially available are relatively benign, there exists a number of pharmaceutical products (some of which are now discontinued due to safety concerns) that have been marketed for off-label use as weight loss aids. The rise in obesity globally, now recognised as a global epidemic by the World Health Organisation (Caballero, 2007) has fuelled both an increase in lifestyle interventions and increasing interest in pharmacological treatments (Milano et al, 2020). An equivalent rise in recent years has been seen in in herbal weight loss products and the use of unlicensed pharmaceuticals, many of which have subsequently been banned due to safety issues (e.g. DMAA, banned for its adverse cardiovascular effects) (Evans-Brown et al, 2012). Generally speaking, weight loss drugs fall into one of two categories; those that suppress appetite and those that increase metabolic rate, usually by stimulant effect. This second group may also suppress appetite.
DNP
 Originally used in the manufacture of explosives in World War I, and subsequently in fungicides, insecticides and photographic development (McVeigh et al, 2016) 2,4-Dinitrophenol (DNP) also has an extensive history as a weight loss drug. Its propensity for causing rapid weight loss was first noted in munitions factories during World War I and led to its development as a patent medicine (drugs available without prescription and, usually, without disclosure of the ingredients). It was, in fact, the first commercially available weight loss drug in the USA (Evans-Brown et al, 2012), widely available throughout the early 1930’s. DNP functions by significantly raising metabolic rate and thereby body temperature, resulting in thermogenesis and subsequent fat loss. However, results are unpredictable and there is no reliable measure by which to calculate appropriate doses. In fact, in terms of dose, the therapeutic window of DNP is extremely small and outside of this the drug either exhibits no tangible effect or potentially serious side-effects (McVeigh et al, 2016). Indeed, it was the growing number of reports of serious adverse effects such as; cataracts, liver failure and multiple fatalities, including some reports of post-mortem temperature rises (McVeigh et al, 2016), that led to the drug being banned in the USA in 1938. Despite the significant potential for adverse effects, the complete lack of reliable human trials and the fact that DNP is only sold as a research chemical, DNP appears to have had a resurgence of interest in recent years (McVeigh et al, 2016). The very high risk of serious adverse effects from DNP, especially given the unknown provenance and dosage of available preparations, underlines the importance of engagement with IPED users, especially those that act as key influencers within the community (McVeigh et al, 2016).
Originally used in the manufacture of explosives in World War I, and subsequently in fungicides, insecticides and photographic development (McVeigh et al, 2016) 2,4-Dinitrophenol (DNP) also has an extensive history as a weight loss drug. Its propensity for causing rapid weight loss was first noted in munitions factories during World War I and led to its development as a patent medicine (drugs available without prescription and, usually, without disclosure of the ingredients). It was, in fact, the first commercially available weight loss drug in the USA (Evans-Brown et al, 2012), widely available throughout the early 1930’s. DNP functions by significantly raising metabolic rate and thereby body temperature, resulting in thermogenesis and subsequent fat loss. However, results are unpredictable and there is no reliable measure by which to calculate appropriate doses. In fact, in terms of dose, the therapeutic window of DNP is extremely small and outside of this the drug either exhibits no tangible effect or potentially serious side-effects (McVeigh et al, 2016). Indeed, it was the growing number of reports of serious adverse effects such as; cataracts, liver failure and multiple fatalities, including some reports of post-mortem temperature rises (McVeigh et al, 2016), that led to the drug being banned in the USA in 1938. Despite the significant potential for adverse effects, the complete lack of reliable human trials and the fact that DNP is only sold as a research chemical, DNP appears to have had a resurgence of interest in recent years (McVeigh et al, 2016). The very high risk of serious adverse effects from DNP, especially given the unknown provenance and dosage of available preparations, underlines the importance of engagement with IPED users, especially those that act as key influencers within the community (McVeigh et al, 2016).
DNP is an oral drug, usually sold in capsule form, but may also be sold in its raw form requiring further preparation by the user. There are no reliable data on appropriate doses, but user forums frequently offer the advice of 2-3mg per kg bodyweight, taken for 7-8 days (McVeigh et al, 2016).
Clenbuterol
 Clenbuterol hydrochloride is a pharmaceutical drug, licenced in some countries as a treatment for asthma (although notably not in the UK or USA). Extensive clinical trials and a long history of use as a therapeutic agent have shown clenbuterol to be well tolerated at therapeutic doses. However, like all stimulant drugs, there are some potential side-effects such as increased blood pressure, palpitations, disrupted sleep patterns and nausea. It should be noted these side-effects tend to resolve as the user acclimatises to the drug but may become more serious at higher doses. As with all IPEDs, it is common to find IPED users taking much higher doses than would be taken therapeutically and as such, there exists a risk of more serious effects on blood pressure, heart arrhythmia, increased anxiety/panic. Clenbuterol is supplied in tablet form, usually in 20mcg doses. A therapeutic dose would be 20-40mcg per day but it is not uncommon for IPED users to use as much as 160mcg (Llewellyn, 2017).
Clenbuterol hydrochloride is a pharmaceutical drug, licenced in some countries as a treatment for asthma (although notably not in the UK or USA). Extensive clinical trials and a long history of use as a therapeutic agent have shown clenbuterol to be well tolerated at therapeutic doses. However, like all stimulant drugs, there are some potential side-effects such as increased blood pressure, palpitations, disrupted sleep patterns and nausea. It should be noted these side-effects tend to resolve as the user acclimatises to the drug but may become more serious at higher doses. As with all IPEDs, it is common to find IPED users taking much higher doses than would be taken therapeutically and as such, there exists a risk of more serious effects on blood pressure, heart arrhythmia, increased anxiety/panic. Clenbuterol is supplied in tablet form, usually in 20mcg doses. A therapeutic dose would be 20-40mcg per day but it is not uncommon for IPED users to use as much as 160mcg (Llewellyn, 2017).
“Fat burners”
Over the counter fat loss preparations, of varying legality, are widely available, typically using stimulants to reduce appetite and increase metabolic rate. Stimulants may include caffeine (at high doses), ephedrine, DMAA (dimethylamylamine, widely banned in sports and banned for OTC sale in the UK, but not illegal at this time), sibutramine (banned, but not illegal) and rimonabant, among other drugs. As with all stimulant drugs, users can experience anxiety, palpitations and sleep disturbance and long-term use carries the potential of adverse psychiatric effects.
Other weight loss drugs
Thyroid drugs, oral preparations prescribed to treat hypothyroidism (levothyroxine (T4) and triiodothyronine (T3)) may be used alongside anabolic agents to facilitate weight loss. The use of these drugs produces a stimulant-like effect and carries much the same risk as stimulant drugs.
Human growth hormone (hGH) and human growth hormone fragment (hGH Fragment 177-191) are sometimes used to facilitate fat loss. Both are injectable peptides and require reconstitution by the end user with sterile water before injection. Human growth hormone fragment is simply one small section of the HGH peptide chain that has been shown to increase lipolysis (Jiang et al, 2001).
Section B: Skin Appearance
Melanotan
 Melanotan was developed in the 1980’s, first as a possible tanning agent that did not require exposure to sunlight (mealnotan I) and then, in a subsequent variation (melanotan II) as a possible treatment for sexual dysfunction. The melanotans function as melanocyte receptor agonists and are believed to exert their tanning function by simulating melanocyte stimulating hormone, thereby causing the production of eumelanin (Wikberg & Mutulis, 2008). Melanotan I was subsequently sold and further developed by a pharmaceutical company into a drug called afamelanotide, whilst melanotan II was developed by a separate company into a drug called bremelanotide. Both drugs have completed Phase III clinical trials. Afamelanotide has been licenced for the treatment of phototoxicity associated with Erythropoietic protoporphyria by the EMA in Europe and FDA in the USA under the brand name Scenesse. The drug is designed to be delivered as an implant that sits just under the skin and last approximately two months. As of 2019, bremelanotide has been licenced by the FDA for treatment of Hypoactive Sexual Desire Disorder, with the drug delivered as a subcutaneous injection by the patient themselves. Early trials with both drugs raised concerns over side-effects with both drugs causing some nausea at administration, sometimes accompanied by headaches and in the case of melanotan II (and early developments of bremelanotide) significantly raised blood pressure in some patients.
Melanotan was developed in the 1980’s, first as a possible tanning agent that did not require exposure to sunlight (mealnotan I) and then, in a subsequent variation (melanotan II) as a possible treatment for sexual dysfunction. The melanotans function as melanocyte receptor agonists and are believed to exert their tanning function by simulating melanocyte stimulating hormone, thereby causing the production of eumelanin (Wikberg & Mutulis, 2008). Melanotan I was subsequently sold and further developed by a pharmaceutical company into a drug called afamelanotide, whilst melanotan II was developed by a separate company into a drug called bremelanotide. Both drugs have completed Phase III clinical trials. Afamelanotide has been licenced for the treatment of phototoxicity associated with Erythropoietic protoporphyria by the EMA in Europe and FDA in the USA under the brand name Scenesse. The drug is designed to be delivered as an implant that sits just under the skin and last approximately two months. As of 2019, bremelanotide has been licenced by the FDA for treatment of Hypoactive Sexual Desire Disorder, with the drug delivered as a subcutaneous injection by the patient themselves. Early trials with both drugs raised concerns over side-effects with both drugs causing some nausea at administration, sometimes accompanied by headaches and in the case of melanotan II (and early developments of bremelanotide) significantly raised blood pressure in some patients.
Harm reduction services in the UK first reported IPED clients using melanotan in the mid-2000s (Evans-Brown et al, 2009b), with a rapid increase in the number of presentations across the UK over the next few years. Interestingly, many of those presenting to needle exchanges for equipment to self-administer melanotan were new clients, with no history of using other IPEDs (such as anabolic steroids). A strong global user community for melanotan use developed, with several large user forums dedicated to discussing its use (Evans-Brown et al, 2009b) and there appears to continue to be a thriving market for the original melanotan I & II formulations. This is despite both drugs being discontinued in clinical trials in favour of analogous drugs, due to adverse effects. Some user reports suggest melanotan II may also assist weight loss alongside its ability to increase libido and cause skin tanning, but there is no robust evidence to support this.
Melanotan is primarily available via unlicensed online sources as a freeze-dried powder requiring reconstitution by the user into an injectable solution using bacteriostatic water. As with all unlicensed drugs, especially those sold for injection, there are concerns over the quality of the product, with some limited evidence showing the presence of unidentified impurities (Breindahl et al, 2015). The drug is injected subcutaneously which, as with all injectable IPEDs, raises concerns over both injecting technique and issues of sterility in the reconstitution and storage of the drug. There is a nasal spray version of melanotan II available through a number of online sources, however, user reports show mixed results. There are a large number of websites showing instructions for reconstituting melanotan and offering advice on dosing, but the quality of the instruction and the range of suggested doses varies considerably. Many websites suggest starting with a low dose (often around 0.5mg) to assess tolerance (especially to side-effects such as nausea). While formulations for nasal administration of melanotan has become increasingly available over the last decade, the demand for injectable melanotan remains.
Section B: Ancillary Drugs (For Side Effects)
The use of AAS typically involves complex polypharmacy of both the anabolic agents themselves and a range of ancillary drugs, including drugs for weight loss, recreational psychoactives, analgesics, stimulants (for training purposes) and prescription drugs re-purposed to mitigate the potential side-effects of the anabolic agents (Ip et al 2018, Sagoe et al 2015). Some of these drugs are covered elsewhere here but it is important to note that some drugs may have multiple uses (e.g. Human growth hormone can be used for both its anabolic and its fat-burning properties).
Anti-Oestrogens
Sagoe et al (2015) identified 13 groups of non-steroidal substances commonly employed by AAS users alongside their use of AAS. Of particular interest are the pharmaceutical preparations used to mitigate potential side-effects of AAS use. Primarily, these are the anti-oestrogens, drugs originally prescribed for use in cancer and/or fertility treatment. The drugs are used for one of three aims; to reduce (or prevent) the conversion of testosterone to oestrogen (aromatase inhibitors such as anastrozole), to selectively block the action of oestrogen in breast tissue (Selective Estrogen Receptor Modulators (SERMs) such as tamoxifen) and to stimulate endogenous testosterone production following an AAS cycle (tamoxifen, clomiphene, hCG).
The use of these drugs to restore natural testosterone production, commonly referred to as post-cycle therapy (PCT), is somewhat controversial, generating extensive debates on user forums as to whether it is necessary and if so, what drugs and dose protocols should be employed. Equally, scientific literature on the treatment of hypogonadism following AAS use is somewhat equivocal and may be confounded by those users who recover naturally (McBride & Coward, 2016) and wide variation in individual reactions to AAS, including dose and duration of cycle (Rahnema et al, 2014). Coward et al (2013) report that hypogonadal males under 50 years old were 10 times more likely to have used anabolic steroids than those over 50, however, not all AAS users in their study reported hypogonadism. Equally, whilst users may recover naturally, it may take as much as two years for full recovery to take place (Tan & Scally, 2009) which is likely to be an uncomfortable experience for the individual, prompting the use of other drugs (besides anabolic agents) to overcome the effects of hypogonadism. Tan & Scally (2009) note that much of the extant literature examining non-clinical AAS use fails to include the post-cycle period, or to recognise the symptoms of hypogonadism (e.g. depression, muscle loss, anxiety, sleep disturbances) perhaps conflating them with symptoms of dependence.
Although there is considerable variation in the exact protocols used, PCT will likely include a combination of tamoxifen (Nolva), clomiphene (Clomid) and hCG (Human Chorionic Gonadotropin). To mitigate oestrogenic side-effects (especially gynaecomastia) whilst on cycle, tamoxifen and/or an aromatase inhibitor such as anastrozole (Arimidex) or exemestane (aromasin) would be taken daily alongside the anabolic agents.
Other Ancillary Drugs
Other drugs (that are not covered elsewhere here) used to mitigate side-effects of AAS use include; erectile dysfunction treatments such as sildenafil or tadalifil, diuretics such as spironolactone or furosemide to mitigate water retention caused by raised oestrogen levels and analgesics, including NSAIDs and opioids, to mitigate pain from training. It should be noted that this is not an exhaustive list, but rather an indication of the potential complexities of the polypharmacy frequently employed by AAS users.
References: Section B
Basaria, S., Collins, L., Dillon, E. L., Orwoll, K., Storer, T. W., Miciek, R., Ulloor, J., Zhang, A., Eder, R., Zientek, H., Gordon, G., Kazmi, S., Sheffield-Moore, M. & Bhasin. S. (2013) The Safety, Pharmacokinetics, and Effects of LGD-4033, a Novel Nonsteroidal Oral, Selective Androgen Receptor Modulator, in Healthy Young Men. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 68.1 pp87-95
Begley, E., J. McVeigh, V. Hope, G. Bates, R. Glass, J. Campbell, C. Tanner, J. Kean, G. Morgan, D. Acreman and J. Smith (2017). Image and Performance Enhancing Drugs: 2016 National Survey Results. Liverpool, Liverpool John Moores University
Brennan, R., Wells, J. G., & Van Hout, M. C. (2014). An unhealthy glow? A review of melanotan use and associated clinical outcomes. Performance Enhancement & Health, 3(2), 78-92. doi:10.1016/j.peh.2015.06.001
Breindahl, T., Evans-Brown, M., Hindersson, P., McVeigh, J., Bellis, M., Stensballe, A. & Kimergård, A. (2015) Identification and characterization by LC-UVMS/MS of melanotan II skin-tanning products sold illegally on the Internet Drug Test. Analysis, 7 pp:164–172
Caballero, B. (2007) The Global Epidemic of Obesity: An Overview Epidemiologic Reviews, 29:1, pp 1–5, https://doi.org/10.1093/epirev/mxm012
Coward, R. M., Rajanahally, S., Kovac, J. R., Smith, R. P., Pastuszak, A. W., & Lipshultz, L. I. (2013). Anabolic steroid induced hypogonadism in young men. The Journal of urology, 190(6), 2200–2205. https://doi.org/10.1016/j.juro.2013.06.010
Dalton J.T., Barnette, K.G., Bohl, C.E., Hancock, M.L., Rodriguez, D., Dodson, S.T., Morton, R.A. & Steiner, M.S. (2011) The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled phase II trial Journal Cachexia Sarcopenia Muscle 2:153–161 DOI 10.1007/s13539-011-0034-6
Evans-Brown, M. & McVeigh, J. (2009) Injecting human growth hormone as a performance-enhancing drug—perspectives from the United Kingdom. Journal of Substance Use, 14:5, 267-288, DOI: 10.3109/14659890903224383
Evans-Brown, M., Dawsom, R.T., Chandler, M. & McVeigh, J. (2009) Use of melanotan I and II in the general population BMJ Feb 17;338:b566. Doi: 10.1136/bmj.b56
Evans-Brown, M., McVeigh, J., Perkins, C., & Bellis, M. A. (2012). Human Enhancement Drugs – The Emerging Challenges to Public Health. Liverpool: Centre for Public Health, Liverpool John Moores University
Flores, J.E., Chitturi, S., & Walker, S. (2020). Drug‐Induced Liver Injury by Selective Androgenic Receptor Modulators. Hepatology Communications. Doi:10.1002/hep4.1456
Google trend search, accessed 23/09/2020: https://trends.google.com/trends/explore?date=all&q=SARMs
Ip, E., Doroudgar, S., Lau, B. & Barnett, M. (2018). Anabolic steroid users’ misuse of non-traditional prescription drugs. Research in Social and Administrative Pharmacy. 15. 10.1016/j.sapharm.2018.07.003.
Jiang WJ., Gianello R., Heffernan M., Ogru E., Libinaki R., Ng F. (2001) Development of a Human Growth Hormone Peptide Analogue AOD9604 into an Anti-Obesity Drug. In: Lebl M., Houghten R.A. (eds) Peptides: The Wave of the Future. American Peptide Symposia, vol 7. Springer, Dordrecht. https://doi.org/10.1007/978-94-010-0464-0_333
Kanno Y, Hikosaka R, Zhang SY, Inoue Y, Nakahama T, Kato K, Yamaguchi A, Tominaga N, Kohra S, Arizono K, Inouye Y. (2011 ) (17α,20E)-17,20-[(1-methoxyethylidene)bis(oxy)]-3-oxo-19-norpregna-4,20-diene-21-carboxylic acid methyl ester (YK11) is a partial agonist of the androgen receptor. Biol Pharm Bull.;34(3):318-23. doi: 10.1248/bpb.34.318. PMID: 21372378.
Kicman AT. (2008 ) Pharmacology of anabolic steroids. Br J Pharmacol. Jun;154(3):502-21. doi: 10.1038/bjp.2008.165. PMID: 18500378; PMCID: PMC2439524.
Kimergard, A., Breindhal, T., Hindersson, P. & McVeigh, J. 2014. The composition of anabolic steroids from the illicit market is largely unknown: implications for clinical case reports. QJM-An International Journal of Medicine, 107, 597-598.
Llewellyn, W. (2017) Anabolics: 11th ed. Molecular Nutrition: Florida
McBride, J. A., & Coward, R. M. (2016). Recovery of spermatogenesis following testosterone replacement therapy or anabolic-androgenic steroid use. Asian journal of andrology, 18(3), 373–380. https://doi.org/10.4103/1008-682X.173938
Milano, W., De Biasio, V., Di Munzio, W., Foggia, G. & Capasso, A. (2020) Obesity: the New Global Epidemic. Pharmacological Treatment, Opportunities and Limits for Personalized Therapy Endocrine, Metabolic & Immune Disorders Drug Targets May DOI: 10.2174/1871530320666200515112853
Murphy, M. G., Plunkett, L. M., Gertz, He, W., Wittreich, J., Polvino, W. M.,& Clemmons, D. R. (1998) MK-677, an Orally Active Growth Hormone Secretagogue, Reverses Diet-Induced Catabolism Journal of Clinical Endocrinology & Metabolism, 83:2, pp320–325, https://doi.org/10.1210/jcem.83.2.4551
Olson EJ, Pearce GL, Jones NP, Sprecher DL. (2012) Lipid effects of peroxisome proliferator-activated receptor-δ agonist GW501516 in subjects with low high-density lipoprotein cholesterol: characteristics of metabolic syndrome. Arterioscler Thromb Vasc Biol. Sep;32(9):2289-94. doi: 10.1161/ATVBAHA.112.247890. Epub 2012 Jul 19. PMID: 22814748.
Rahnema, C. D., Lipshultz, L. I., Crosnoe, L. E., Kovac, J. R., & Kim, E. D. (2014). Anabolic steroid-induced hypogonadism: diagnosis and treatment. Fertility and sterility, 101(5), 1271–1279. https://doi.org/10.1016/j.fertnstert.2014.02.00
Sagoe, D., McVeigh, J., Bjørnebekk, A. et al. (2015) Polypharmacy among anabolic-androgenic steroid users: a descriptive metasynthesis. Subst Abuse Treat Prev Policy 10, 12. https://doi.org/10.1186/s13011-015-0006-5
Sigma Aldrich Handling and Storage of Synthetic Peptides https://www.sigmaaldrich.com/content/dam/sigma-aldrich/docs/Sigma/General_Information/peptide_handling_guide.pdf Accessed: 05/11/2020
Solomon, Z.J., Mirabal, J.R., Mazur, D.J., Kohna, T.P., Lipshultz, L.I. & Pastuszak, A.W. (2019) Selective Androgen Receptor Modulators (SARMs) – Current Knowledge and Clinical Applications Sex Med Rev. 7(1): 84–94. Doi:10.1016/j.sxmr.2018.09.006
Tan, R. S., & Scally, M. C. (2009). Anabolic steroid-induced hypogonadism–towards a unified hypothesis of anabolic steroid action. Medical hypotheses, 72(6), 723–728. https://doi.org/10.1016/j.mehy.2008.12.042
Van Hout, M. C., & McVeigh, J. (2019). The use of sunless synthetic tanning products. In K. Van de Ven, K. Mulrooney, & J. McVeigh (Eds.), Human enhancement drugs. Abingdon, Oxon: Routledge.Wikberg JE, Mutulis F. (2008) Targeting melanocortin receptors: an approach to treat weight disorders and sexual dysfunction. Nat Rev Drug Discov 7:307-23.
Yu Z, He S, Wang D, Patel H.K., Miller C.P., Brown J.L., Hattersley G., Saeh J.C. (2017). Selective Androgen Receptor Modulator RAD140 Inhibits the Growth of Androgen/Estrogen Receptor-Positive Breast Cancer Models with a Distinct Mechanism of Action. Clinical Cancer Research. 23 (24): 7608–7620. Doi:10.1158/1078-0432.CCR-17-0670

Section C: Prevalence
Whilst there is some evidence to suggest a significant rise in self-directed IPED use, especially since the 1990’s (McVeigh & Begley, 2016; Pope et al, 2014; Sagoe & Pallesen, 2018), there are currently no reliable measures of prevalence. Korkia & Stimpson (1997) reported the prevalence of AAS at any time was 9.1% for males and 2.3% for females, based on a sample of 1,667 participants in gymnasia across England, Scotland & Wales. However, they found wide variability across the gymnasia that took part in the survey, from no reports in some, up to 46% in others. This highlights a fundamental issue in calculating prevalence of drug use that is either illicit, or at least occupies a legal grey area (depending on the location); the numbers will reflect the particular groups surveyed, but may not be applicable to the wider population. In the UK for example, the Crime Survey for England & Wales (CSEW) provides figures for the use of anabolic steroids and reports that, for the year ending March 2019, prevalence of lifetime AAS for 16-59 year olds was 1.1%, whilst past year was just 0.2% for the same group (ONS, 2019). However, as noted by the Advisory Council on the Misuse of Drugs (ACMD), the survey suffers severe limitations; it relies on self-reported behaviour, by a traditionally closed sub-culture, via a survey with limited distribution (ACMD, 2010, p17). Other work in the UK reports an increase in presentations for AAS use in needle exchanges in the northwest between 1995 and 2015; with AAS clients representing 54.9% of all clients in 2015, up from 17.1% in 1995 (McVeigh & Begley, 2016). Prevalence estimates from this study, based on male users aged 20-29, reveals substantial increases in six of the eight local authority areas sampled, with the two remaining areas showing much smaller changes, one of which was a small decrease. There was wide variability across the areas sampled from 1.17 per 1,000 population to 18.7. A further study of needle exchanges across the north of England reports the proportion of needle exchange clients presenting for AAS use across a wider geographic area of 14 local authority areas (including those in McVeigh & Begley, 2016). Results again showed wide variability, from 34% to 86% of all clients presenting for AAS use (Kimergard & McVeigh, 2014).
Outside of the UK, there are a number of studies exploring prevalence. Pope et al (2014) estimated between 2.9 – 4 million Americans had used AAS at some point in their lifetime, with approximately one million experiencing AAS dependence (Pope et al, 2014 p374), based on analysis of pooled youth and household surveys. Related; Kanayama & Pope (2018) highlight some of the inherent issues in such survey instruments, especially with regard to the wording of questions relating to anabolic steroids that may cause false-positives in responses and report the wide variability in prevalence rates both between and within countries. They further highlight that, whilst many believe the issue of self-directed AAS use is primarily a doping issue amongst competing athletes, in fact the vast majority of people using AAS do not report using them to improve competitive performance (Kanayama & Pope, 2018 p9). In fact, the World Anti-Doping Agency (WADA) consistently reports between just 1-2% positive adverse analytical findings doping tests, although this has been challenged by other work suggesting the actual figure is much higher (Ulrich et al, 2018). Sagoe & Pallesen (2018) report that prevalence of AAS use is higher in Western cultural contexts, the Middle East and South America and lower in Africa and Asia (Sagoe & Pallesen, 2018, p9) and highlights the higher prevalence amongst particular sub-groups (males, athletes, injecting drug users, recreational athletes, and sexual and gender minority groups) and there is further evidence of rising prevalence in the Eastern Mediterranean (Hearne et al, 2020).
Whilst there are clear issues with calculating prevalence of AAS use, there exists a growing body of evidence to show a rise in presentations to health services by IPED users and a growing and widespread availability of AAS and other IPEDs (Brennan et al, 2017, McBride et al, 2018). Coupled with the apparent rise in novel IPEDs such as SARMs (Solomon et al, 2019) and growing evidence of harms associated with IPED use it is crucial that more efforts are made to establish reliable prevalence measures if we are to measure the impact of health interventions.
References: Section C
ACMD. (2010) Advisory Council on the Misuse of Drugs, Consideration of the Anabolic Steroids. London: Home Office.
Brennan, R., Wells, J.S.G. & Van Hout, M.C. (2017) The injecting use of image and performance-enhancing drugs (IPED) in the general population: a systematic review Health and Social Care in the Community 25(5), pp1459–1531
Hearne, E., Wazaify, M., VanHout, M.C., Atkinson, A. & McVeigh, J. (2020) Anabolic-Androgenic Steroid Use in the Eastern Mediterranean Region: a Scoping Review of Extant Empirical Literature International Journal of Mental Health and Addiction. 1-28. 10.1007/s11469-019-00217-8.
Kanayama, G. & Pope, H.G. (2018) History and epidemiology of anabolic androgens in athletes and nonathletes. Molecular and Cellular Endocrinology 464 pp:4-13
Kimergård, A. & McVeigh, J. (2014) Variability and dilemmas in harm reduction for anabolic steroid users in the UK: a multi-area interview study Harm Reduction Journal 11:19
Korkia, P., Stimson, G. V. (1997) lndications of Prevalence, Practice and Effects of Anabolic Steroid Use in Great Britain. Int. J. Sports Med., Vol. 18, pp. 557 – 562
McBride, J. A., Carson, C.C. & Coward, R.M. (2018) The Availability and Acquisition of Illicit Anabolic Androgenic Steroids and Testosterone Preparations on the Internet American Journal of Men’s Health Vol. 12(5) pp1352–1357
McVeigh, J. & Begley, E. (2016) Anabolic steroids in the UK: an increasing issue for public health Drugs: Education, Prevention and Policy, DOI:10.1080/09687637.2016.1245713
Office for National Statistics: Drugs Misuse: Findings from the 2018/19 Crime Survey for England and Wales www.gov.uk/government/statistics/drug-misuse-findings-from-the-2018-to-2019-csew
Pope, H.G., Kanayama, G., Athey, A., Ryan, E., Hudson, J.I. & Baggish, A. (2014) The Lifetime Prevalence of Anabolic-Androgenic Steroid Use and Dependence in Americans: Current Best Estimates Am J Addict Jul 23(4) pp371-377 doi: 10.1111/j.1521-0391.2013.12118.x
Sagoe, D. & Pallesen, S. (2018) Androgen abuse epidemiology Curr Opin Endocrinol Diabetes Obes 25 DOI:10.1097/MED.0000000000000403
Solomon, Z.J., Mirabal, J.R., Mazur, D.J., Kohna, T.P., Lipshultz, L.I. & Pastuszak, A.W. (2019) Selective Androgen Receptor Modulators (SARMs) – Current Knowledge and Clinical Applications Sex Med Rev. 7(1): 84–94. Doi:10.1016/j.sxmr.2018.09.006
Ulrich, R., Pope Jr.H.G., Cleret, L. Petroczi, A., Nepusz, T., Schaffer, J., Kanayama, G., Comstock, R.D. & Simon, P. (2018) Doping in Two Elite Athletics Competitions Assessed by Randomized-Response Surveys Sports Med 48:211–219 https://doi.org/10.1007/s40279-017-0765-4

Section D: Characteristics of users
The development of effective health interventions and service provision for AAS users ideally requires a robust understanding of user characteristics, but research in this area is limited. Early work in the UK identified a simple categorisation of AAS users comprising; ‘competitive sports participants’ (people using AAS to enhance performance, including both professional and amateur athletes), ‘occupational users’ (people using AAS to enhance physical ability or appearance related to their job, e.g. doormen, actors etc) and ‘recreational users’ (those using AAS for primarily image related reasons) (Lenehan et al, 1996). Whilst these categories provided a reasonable model of user characteristics at the time, they are limited in both scope and detail (Zahnow et al, 2018). Furthermore, the subsequent increase in the number of people presenting to IPED-related services (McVeigh & Begley, 2016) would suggest a need to refine and develop our understanding of IPED users to facilitate development of tailored services and harm minimisation interventions that best meet the needs of this complex and heterogeneous group.
For instance; much of the extant research focuses on AAS use amongst males, especially younger males within the gym environment, often with a bodybuilding focus (either competitive or recreational). Whilst males undoubtedly form the largest group of users, the available evidence also shows female use, albeit on a much smaller scale (Lenehan et al 1996, Korkia & Stimpson 1997, Begley et al 2017). Female users can experience many of the same harms as their male counterparts but there are also a range of side-effects of AAS use that are unique to women, in particular, the masculinising effects of virilisation (ACMD, 2010, Onakomaiya & Henderson 2016) and some evidence to show different patterns of drug use amongst female IPED users (Sverkersson et al, 2020, Begley et al 2017, Havnes et al, 2020c).
More recent work suggests a more detailed typology of users (Christiansen et al 2016) from a series of interviews with 37 male AAS users in Denmark, based on the user’s approach to the risks and effectiveness of use. The types were defined as; the “expert type”, the “YOLO” (You Only Live Once), the “athlete type” and the “wellbeing type”. A similar grouping was found in more recent work by Zahnow et al (2018) using cluster analysis of 611 male participants in a UK study. These typologies serve as a framework through which to understand the diversity of motivations and practices of some AAS users but are not intended as rigid model into which all users will fit. It is important to note that, whilst a specific user may exhibit attributes predominantly associated with a particular typology at one point in time, they may also exhibit traits from other types, or move between types over time (Christiansen et al, 2016).
Beyond these typologies, we can identify other groups for whom AAS use may be more prevalent and/or present specific issues, requiring a more tailored approach in interventions and services. For instance, Ip et al (2019) report lifetime prevalence of AAS use of 21% amongst a sample of 153 gay and bisexual men. They further report that AAS users in the study were more likely to use other drugs (including psychoactives) and engage in unsafe sexual behaviours than those that did not use AAS. There is evidence of IPED use in institutional populations that, by virtue of the setting, may present unique challenges to the provision of appropriate services, such as the military (Kao et al 2020, Taylor et al 2017) or prison populations. Havnes et al (2020) reports lifetime prevalence of AAS use at 28.5% amongst a sample of 1,499 prison inmates, with AAS users also a reporting younger debut age for psychoactive drugs and a higher mean number of psychoactive drugs used than non-AAS users. Van Hout & Kean (2015) highlight the use of AAS within British South Asian communities, describing an observed contrast between the motivations and practices of this group as compared to the UK general population, as well as differences between older and younger users, with older users taking a more measured approach to their AAS use. The authors highlight issues in engagement with BME groups by health services, especially drug services, further highlighting the diversity of the IPED community and the complexity of delivering appropriate services. The issue of differing age groups is further highlighted by Begley et al (2017) who report similar proportions of males 40 years and over as those under the age of 25 amongst a sample of 684 IPED users. As with other drug users, differing age groups may require more tailored health services and interventions. In the UK, health service provision for IPED users is delivered via harm reduction services, usually through community and specialist needle exchanges. However, not all IPED users inject and such services may therefore fail to engage with, or be able to provide appropriate services for, this sub-group of non-injectors (van de Ven et al, 2019). Equally; services in the UK have reported a novel client group of melanotan injectors, who may not use other IPEDs but are nonetheless part of a seemingly growing group of people using unlicensed research chemicals to achieve specific physical changes (Evans-Brown et al, 2009) and therefore exposed to many of the same risks as other IPED users.
A number of studies point to an association between the use of psychoactive drugs and AAS use, with some studies highlighting greater prevalence of psychoactive drug use amongst AAS users compared to the general population (Havnes et al, 2020a and 2020b, Sagoe et al, 2015). Christiansen et al (2016) noted differences between their typologies, with the ‘YOLO’ group being most likely to use recreational drugs and other groups being opposed to the idea, whilst Zahnow et al (2018) noted variations in both recreational drug use and alcohol use between differing groups of AAS users. These sub-groups of AAS users may therefore require additional health service provision reflecting the broader range of substances used and the potential issues they may raise.
The typologies previously discussed present a framework for modelling health service provision and interventions, but it is important to note that within and across those typologies, there are variations in motivation for IPED use that may be associated with differences in patterns of substance use and/or health issues requiring an appropriately designed range of services. Christiansen et al (2016) note a range of motivations across the typologies, accompanied by differences in substance use behaviour. For instance, the ‘YOLO’ type is motivated by things like impatience and influence from their peers, expresses little concern over possible side-effects of their AAS use, has low levels of knowledge about their drug use and is more likely to use a range of other drugs, including psychoactives. Contrast this with the ‘Expert’ type, who is motivated by ‘lay scientific curiosity’, is knowledgeable, concerned about possible side-effects, monitors their own health, takes a more measured approach to their AAS use and is much less likely to use psychoactive drugs (Christiansen et al, 2016, p9). Similar variation was reported across the clusters identified in Zahnow et al (2018). Murray et al (2016) noted differences between those using AAS primarily for appearance purposes and those using them for performance purposes amongst a sample of 122 males. Those using AAS primarily for appearance enhancement were reported as having greater psychopathology around eating disorders, and muscle dysmorphia and greater ‘drive for size’ (p200). This pattern is also noted in other work (Kanayama et al, 2001) and highlights a possible need for mental health service provision within any intervention.
References: Section D
ACMD. (2010) Advisory Council on the Misuse of Drugs, Consideration of the Anabolic Steroids. London: Home Office.
Begley, E., J. McVeigh, V. Hope, G. Bates, R. Glass, J. Campbell, C. Tanner, J. Kean, G. Morgan, D. Acreman and J. Smith (2017). Image and Performance Enhancing Drugs: 2016 National Survey Results. Liverpool, Liverpool John Moores University.
Christiansen, A. V., A. S. Vinther and D. Liokaftos (2016). Outline of a typology of men’s use of anabolic androgenic steroids in fitness and strength training environments. Drugs: Education, Prevention and Policy 24(3) pp295-305.
Evans-Brown, M., Dawsom, R.T., Chandler, M. & McVeigh, J. (2009) Use of melanotan I and II in the general population BMJ Feb 17;338:b566. Doi: 10.1136/bmj.b56
Havnes, I.A., Bukten, A., Rognli, E.B. & Muller, A.E. (2020) Use of anabolic-androgenic steroids and other substances prior to and during imprisonment – Results from the Norwegian Offender Mental Health and Addiction (NorMA) study Drug and Alcohol Dependence Volume 217, 1 December
Havnes, I.A, Jørstad, M.L., McVeigh, J., Van Hout, M.C., & Bjørnebekk, A. (2020b) The Anabolic Androgenic Steroid Treatment Gap: A National Study of Substance Use Disorder Treatment Substance Abuse: Research and Treatment Volume 14: 1–9
Havnes, I. A., Jorstad, M. L., Innerdal, I., & Bjornebekk, A. (2020). Anabolic-androgenic steroid use among women – A qualitative study on experiences of masculinizing, gonadal and sexual effects. Int J Drug Policy, 102876. doi:10.1016/j.drugpo.2020.102876.
Kanayama, G., Pope Jr., H.G., Hudson, J.I., (2001) ‘Body image’ drugs: a growing psychosomatic problem. Psychother. Psychosom. 70, 61–65, http://dx.doi.org/10.1159/000056228.
Kao, T.C, Kazman, J.B., Cheng, Y.H., Jeffery, D.D. & Deuster, P.A. (2020) Healthy Lifestyles Among Military Active Duty Service Members, and Associations With Body Building And Weight Loss Supplement Use Annals of Epidemiology https://doi.org/10.1016/j.annepidem.2020.08.011
Korkia, P. & Stimson, G. V. (1997) lndications of Prevalence, Practice and Effects of Anabolic Steroid Use in Great Britain. Int. J. Sports Med., Vol. 18, pp. 557 – 562
Lenehan, P., Bellis, M., & McVeigh, J. (1996). A study of anabolic steroid use in the North West of England. Journal of Performance Enhancing Drugs, 1.
Murray, S.B. Griffiths, S., Mond, J.M., Kean, J. & Blashill, A.J. (2016) Anabolic steroid use and body image psychopathology in men: Delineating between appearance- versus performance-driven motivations Drug and Alcohol Dependence 165 pp198–202
Onakomaiya, M.M. & Henderson, L.P. (2016) Mad men, women and steroid cocktails: A review of the impact of sex and other factors on anabolic androgenic steroids effects on affective behaviors Psychopharmacology (Berl). 2016 February ; 233(4): 549–569. doi:10.1007/s00213-015-4193-6.
Sagoe, D., McVeigh, J., Bjørnebekk, A., Essilfie, M.S., Andreassen, C.S. & Pallesen, S. (2015) Polypharmacy among anabolic-androgenic steroid users: a descriptive metasynthesis Substance Abuse Treatment, Prevention, and Policy 10:12 doi: 10.1186/s13011-015-0006-5
Sverkersson, E., Andreasson, J. & Johansson, T. (2020) ‘Sis Science’ and Fitness Doping: Ethnopharmacology, Gender and Risk Soc. Sci. 9, 55; doi:10.3390/socsci9040055
Zahnow, R., McVeigh, J., Bates, G., Hope, V., Kean, J., & Campbell, J. (2018). Identifying a typology of men who use Anabolic Androgenic Steroids (AAS). The International Journal of Drug Policy, 55 pp105–112
Van Hout, M.C. & Kean, J. (2015) An exploratory study of image and performance enhancement drug use in a male British South Asian community International Journal of Drug Policy 26 pp860–867
van de Ven, K. Zahnow, R., McVeigh, J. & Winstock, A. (2019): The modes of administration of anabolic-androgenic steroid users (AAS): are noninjecting people who use steroids overlooked? Drugs: Education, Prevention and Policy DOI:10.1080/09687637.2019.1608910

Section E: Harms
Background: The use of IPEDs is associated with a diverse range of physical and psychological harms that can vary widely according to substance, dose, duration of use, route of administration, drug combinations and idiosyncratic reactions. However, whilst there exists a strong body of evidence to show association between the use of IPEDs (especially AAS) and adverse health effects, the literature is less clear on the issue of causality (ACMD, 2010). Some physical effects (e.g. acne, gynaecomastia, hypogonadism) are sufficiently commonly reported to suggest a causal link, but others (e.g. cardiovascular disease, issues around liver function, adverse psychological effects) are less commonly reported and difficult to establish a causal link for (Kanayama et al, 2008). Determining causal links between IPED use and adverse health effects is confounded by a number of issues. The vast majority of the extant literature are case studies, animal studies, cross-sectional or clinical trials with therapeutic doses (Evans-Brown et al, 2009). The small number of studies employing doses typically seen in self-directed use amongst bodybuilders and similar groups, lack the complex polypharmacy often employed by IPED users and avoid the potential harms associated with poor quality products that can often be found in general use, as well as the sometimes lengthy durations of use and more extreme doses sometimes employed. Conversely, randomised clinical trials that could address many of these issues would be ethically unsound and therefore unlikely to be undertaken (Pope et al, 2014). Beyond the effects of the drugs themselves, the route of administration may also present unique risks. Poor injecting practice can lead to the formation of abscesses at the injection site and the potential transmission of blood borne viruses. Hope et al (2016) report that prevalence of blood borne viruses (HIV, Hep B, Hep C) have increased amongst UK IPED injectors, with the prevalence for HIV in particular, now similar to that found amongst injectors of psychoactive drugs.
The physical and psychological harms commonly associated with AAS use (and associated ancillary drugs) are briefly described in Table 3; more information on these can be found at https://www.ipedinfo.co.uk/ipeds-risks-and-complications/. For many of the physical harms listed here, it is common for users to attempt to mitigate these effects by using ancillary drugs.
Table 3: Commonly reported adverse effects of AAS use
| Gynaecomastia | Development of breast tissue in males, severe cases require surgery to correct. |
| Endocrine & Genitourinary | Reduction in testicle size and function, likely leading to adverse effects on natural testosterone production and fertility. Recovery may take several months following cessation of AAS use. |
| Acne | Increase in acne on face, chest and back. Some case reports of more severe cases (acne conglobata) requiring treatment. |
| Male Pattern Baldness (androgenic alopecia) | Commonly associated with AAS derived from DHT, may simply exacerbate existing predisposition rather than being directly causal. |
| Hypertension | Increased blood pressure whilst using AAS is commonly reported, but current data are equivocal on causality. Reports suggest levels may return to normal following cessation of use. |
| Liver function | Commonly associated with the use of oral AAS, with some case studies reporting acute liver injury. Data on this are equivocal however and it is important to note that muscle damage caused by heavy resistance exercise can cause similar markers to liver damage. |
| Lipid metabolism | Use of AAS is associated with a decrease in HDL and a possible increase in LDL. Severity of effect appears to be linked to the type of steroid used, route of administration, duration of use and may of course, be exacerbated by diet and other lifestyle factors. Reports suggest that lipid metabolism can return to normal following cessation of use, but again, data are equivocal as to how long this may take. |
| Cardiovascular | Recent studies have begun to build a compelling evidence base for the effects of AAS use on cardiac structure and function, with several studies reporting left ventricular hypertrophy and cardiomyopathy, characterised especially by reduced ejection fraction. The exact relationship between AAS use and these effects however, remains unclear. (See below for more detail) |
| Virilisation (in female AAS users) | Virilisation is the development of male characteristics in females and may include hirsutism, deepening of the voice, clitoral enlargement, atrophy of breast tissue, & amenorrhoea. Even relatively low doses of some AAS may produce these effects and at least in some cases, may be permanent. The risks are difficult to elucidate as data on female AAS use are extremely limited. |
| Psychological & Behavioural | Whilst numerous case studies have linked AAS use to a range of psychological and/or behavioural effects such as mania, aggression & depression, the data suggest the effects can be highly idiosyncratic. There is some limited evidence to suggest higher doses increase the likelihood of adverse psychological effects, but again the data are equivocal. |
| Dependence | Whilst there are some studies suggesting AAS may cause psychological dependence in some users, they are primarily based on self-report measures and limited in scope and number and as such, there are little data available. |
Harms associated with AAS use: Emerging Evidence
Cardiovascular effects of AAS use
High doses of AAS have been linked with a range of cardiovascular conditions, including (but not limited to); left ventricular hypertrophy, hypertension, dyslipidaemia, myocardial infarction, arrhythmia, atherosclerosis and cardiomyopathy (Kanayama et al, 2008, Pope et al 2014). Whilst the evidence base remains limited, emerging data show greater cardiac mass amongst AAS users (Far et al, 2012) and evidence of significantly reduced ejection fraction and higher coronary artery plaque volume among current AAS users, as compared to non-users or those currently off cycle (Baggish et al, 2017). The exact mechanisms remain under investigation, but the data suggest that higher doses (especially over 1,000mg per week) and longer durations of use may significantly increase the risk of adverse cardiovascular effects (Baggish et al, 2017, Pope et al, 2014).
Brain Structure
Little is known about the effects of AAS on brain structure, especially at the supraphysiologic doses often employed by AAS users. However, AAS are able to pass the blood-brain barrier and can directly affect the central nervous system (Bjørnebekk et al, 2017) so it would seem logical to suggest that there may be some structural impact that may lead to, or exacerbate, other issues such as psychological side-effects. Kanayama et al (2013) report that AAS users performed significantly more poorly than non-users on tests of visuospatial memory and performance declined with increasing lifetime AAS dose. Kaufman et al (2015) noted significant structural differences in the brains of 10 AAS users vs 10 matched, non-AAS using weightlifters that could lead to increased risk of neurodegenerative processes. A more recent study by Bjørnebekk et al (2017) reports similar differences in brain structure amongst a larger sample of 86 of AAS users compared to 68 non-users. Importantly, the results of all these studies held after controlling for possible confounding factors, suggesting a possible causal link between AAS use and structural alterations to the brain. This therefore lends weight to the possibility of adverse cognitive effects from long-term and/or heavy AAS use, although considerably more research is needed to elucidate the relationship.
Infection
Whilst the use of AAS and other IPEDs appears to carry risk of adverse effects form the drugs themselves, it is important to note that there exists a range of secondary risks from other behaviours associated with AAS use. The vast majority of AAS are injected and as such, carry associated risks from poor injecting practice. Injecting AAS users are particularly vulnerable to localised infection from contaminated products or unsterile injecting processes (Hope et al (2013). Equally, whilst there does not appear to be a high level of needle sharing amongst AAS users (Pope et al, 2014) there is an emerging body of evidence to show a higher prevalence of blood borne viruses amongst AAS users than the general population. Hope et al (2013) HIV prevalence amongst 395 male AAS users of 1.5% (0.8% when adjusted for men who have sex with men or men who inject psychoactive drugs), a figure similar to that found amongst psychoactive drug injectors in England & Wales (1.2%). The same study found lower prevalence levels of Hepatitis B and C ((8.8% and 5.5% respectively) than amongst psychoactive drug injectors, but still higher than the general population. These findings were repeated in Hope et al (2016) where needle sharing was also reported, albeit at comparatively low levels compared to psychoactive drug injectors. Further work by Hope et al (2017) shows approximately one in twenty AAS injectors have hepatitis C, but amongst those who did not also inject psychoactive drugs, the HCV infection was mostly undiagnosed due to poor uptake of testing in this group.
It is important to note that the samples used in these studies may not be representative of the larger IPED community. Participants were recruited via needle exchanges in England & Wales and as such, may not be representative of those who do not access harm reduction services. Furthermore, the prevalence of blood borne viruses is linked, at least in part, to high risk sexual behaviours and psychoactive drug use and as such, there may be a need for targeted services aimed at these higher risk AAS users. Regardless, there is evidence of blood borne viruses (BBV) and unsafe injecting practices amongst a small group of AAS users who do not inject other (psychoactive) drugs and further research is required in this area to determine the scope of the problem.
One possible confounding issue with research in this area is the high levels of secondary needle and syringe distribution in the UK. Glass et al (2019) report on a sample of 537 IPED injectors; 87% accessed needle exchange services and of those, 34% reported collecting injecting equipment for others. In total, 154 participants reported collecting injecting equipment for between 639-1569 people. Whilst increased levels of needle and syringe distribution are welcome, high levels of secondary distribution may mean a large proportion of the IPED community are not accessing harm reduction, thereby impacting the provision of health services, including BBV testing.
Issues of drug quality
As previously stated, much of the extant literature on harms associated with IPED use relies on cross-sectional and/or observational or self-report data. Countering this with more robust study designs, such as randomised clinical trials, is fraught with ethical and logistical issues that are unlikely to be overcome (Evans-Brown et al, 2009). Consequently, much of our knowledge in this area is based on the use of drugs of unknown provenance, the quality of which may impact outcomes such as adverse side-effects (Kimergard et al, 2014). Graham et al (2009) report analytical findings from a study of 57 injectable or oral AAS. Fifty-three percent of the injectable products and 21% of the orals were other than as described on their label, either containing a different active ingredient or no active ingredient at all, a finding supported by other similar studies (Evans-Brown, 2009). Beyond the illicitly sourced drugs themselves, many AAS users will also use a range of over-the-counter dietary sports supplements that may themselves be contaminated. Evans-Brown (2014) note the presence of the breast cancer drug tamoxifen in an over-the-counter product aimed at those using anabolic agents and similar findings have been reported in a number of other studies showing the presence of anabolic agents or other IPEDs in dietary supplements that are available over-the-counter (Cho et al, 2015, Van Poucke et al 2007). Such supplements of course, may not just increase the risk for those already using IPEDs, but also for those who do not use IPEDs but are using these dietary products without being aware of the contents.
The key issue here is that, whilst there is a small evidence base to suggest that a proportion of IPEDs and dietary supplements in common use may be something other than as labelled and/or bacterially contaminated, the extent of the problem is unknown. Further research is required in this area to determine the scope of the problem but also, future research into the effects and side-effects of AAS use must adjust for potential confounding caused by drug quality.
References: Section E
ACMD. (2010) Advisory Council on the Misuse of Drugs, Consideration of the Anabolic Steroids. London: Home Office.
Baggish A.L., Weiner R.B., Kanayama G, Hudson, J.I., Lu, M.T., Hoffman, U. & Pope, H.G. (2017) Cardiovascular toxicity of illicit anabolic-androgenic steroid use. Circulation; 135(21):1991–2002.
Bjørnebekk, A. Walhovd, K.B. Jørstad, M.L., Due-Tønnessen, P., Hullstein, I.R. & Fjell, A.M. (2017) Structural Brain Imaging of Long-Term Anabolic-Androgenic Steroid Users and Nonusing Weightlifters Biological Psychiatry August 15; 82:294–302
Cho SH, Park HJ, Lee JH, Do JA, Heo S, Jo JH, Cho S. (2015) Determination of anabolic-androgenic steroid adulterants in counterfeit drugs by UHPLC-MS/MS. J Pharm Biomed Anal.;111:138-46. doi: 10.1016/j.jpba.2015.03.018. Epub 2015 Mar 28. PMID: 25880245.
Evans-Brown M, Kimergård A, McVeigh J. Elephant in the room? The methodological implications for public health research of performance-enhancing drugs derived from the illicit market. (2009) Drug Test Anal. Jul;1(7):323-6. doi: 10.1002/dta.74. PMID: 20355210.
Evans-Brown, M., McVeigh, J., Perkins, C., & Bellis, M. A. (2012) Human enhancement drugs: The emerging challenges to public health. Liverpool: Public Health Observatories in England
Evans-Brown M, Kimergård A, McVeigh J, Chandler M, Brandt SD. (2014) Is the breast cancer drug tamoxifen being sold as a bodybuilding dietary supplement? BMJ. Feb 13;348:g1476. doi: 10.1136/bmj.g1476. PMID: 24524927.
Far HR, Ågren G, Thiblin I. (2012) Cardiac hypertrophy in deceased users of anabolic androgenic steroids: an investigation of autopsy findings. Cardiovasc Pathol.;21(4): 312–316.
Glass R, Hope VD, Njoroge J, Edmundson C, Smith J, McVeigh J, Parry J, & Desai M. (2019) Secondary distribution of injecting equipment obtained from needle and syringe programmes by people injecting image and performance enhancing drugs: England and Wales, 2012-15. Drug Alcohol Depend. Feb 1;195:40-44. doi: 10.1016/j.drugalcdep.2018.11.021. Epub 2018 Dec 14. PMID: 30580202.
Graham MR, Ryan P, Baker JS, Davies B, Thomas NE, Cooper SM, Evans P, Easmon S, Walker CJ, Cowan D, Kicman AT. (2009) Counterfeiting in performance- and image-enhancing drugs. Drug Test Anal. Mar;1(3):135-42. doi: 10.1002/dta.30. PMID: 20355187.
Hope VD, McVeigh J, Marongiu A, Evans-Brown, M., Smith, J. Kimergård, A., Croxford, S., Beynon, C.M., Parry, J.V., Bellis, M.A. & Ncube, F. (2013) Prevalence of, and risk factors for, HIV, hepatitis B and C infections among men who inject image and performance enhancing drugs: a cross-sectional study BMJ Open 3:e003207. doi:10.1136/bmjopen-2013-003207 1
Hope VD, Harris R, McVeigh J, Cullen KJ, Smith J, Parry JV, DeAngelis D, & Ncube F. (2016) Risk of HIV and hepatitis B and C over time among men who inject image and performance enhancing drugs in England and Wales: Results from cross-sectional prevalence surveys, 1992-2013 Journal of Acquired Immune Deficiency Syndromes, 71 :331-337 doi: 10.1097/QAI.0000000000000835
Hope VD, McVeigh J, Smith J, Glass R, Njoroge J, Tanner C, Parry JV, Ncube F, Desai M. (2017) Low levels of hepatitis C diagnosis and testing uptake among people who inject image and performance enhancing drugs in England and Wales, 2012-15. Drug Alcohol Depend. Oct 1;179:83-86. doi: 10.1016/j.drugalcdep.2017.06.018. Epub 2017 Jul 18. PMID: 28759821.
Kanayama, G., Hudson, J.I. & Pope, H.G. (2008) Long-term psychiatric and medical consequences of anabolic–androgenic steroid abuse: A looming public health concern? Drug and Alcohol Dependence 98 pp1–12
Kanayama G, Kean J, Hudson JI, & Pope HG Jr (2013): Cognitive deficits in long-term anabolic-androgenic steroid users. Drug Alcohol Depend 130:208–214.
Kaufman MJ, Janes AC, Hudson JI, Brennan BP, Kanayama G, Kerrigan AR,Jensen, E. & Pope, H.G. (2015): Brain and cognition abnormalities in long-term anabolic-androgenic steroid users. Drug Alcohol Depend 152:47–56.
Kimergård A, Breindahl T, Hindersson P, & McVeigh J. (2014) The composition of anabolic steroids from the illicit market is largely unknown: implications for clinical case reports. QJM. Jul;107(7):597-8. doi: 10.1093/qjmed/hcu101. Epub 2014 May 7. PMID: 24811551.
Pope, H.G., Wood, R.I., Rogol, A., Nyberg, F., Bowers, L. & Bhasin, s. (2014) Adverse Health Consequences of Performance-Enhancing Drugs: An Endocrine Society Scientific Statement Endocrine Reviews June 35(3):341–375
Van Poucke C, Detavernier C, Van Cauwenberghe R, Van Peteghem C. (2007) Determination of anabolic steroids in dietary supplements by liquid chromatography-tandem mass spectrometry. Anal Chim Acta. Mar 14;586(1-2):35-42. doi: 10.1016/j.aca.2006.09.050. Epub 2006 Oct 4. PMID: 17386694.

Section F: Effective harm reduction & treatment for IPED users
IPED use presents complex challenges for health services and research in effective interventions and services is still at a very early stage. People who use IPEDs are a diverse community with many potential sub -groups requiring targeted interventions, such as men who have sex with men, female users, older users and people who also use psychoactive drugs (especially those who inject) amongst others. People may of course belong to more than one of these groups and/or move between them over time. Providing effective services for these groups therefore likely requires substantial training for health care workers, in order to understand the possible issues this diverse population may experience associated with their drug use, as well as (potentially) access to a wide range of specialist support services (i.e. phlebotomy, endocrinology, mental health and others). Furthermore, such training would ideally require regular updating in order to address potential issues with novel and emerging drugs, as evidence shows the range of drugs and patterns of use evolve (McVeigh et al, 2020).
There are limited data available on effective service provision for IPED users; in part because there are limited services available but also because IPED users are reluctant to engage with health services for issues related to their IPED use (Zahnow et al, 2017) possibly due to actual or perceived stigma (Yu et al, 2015, Harvey et al. 2019, Hope et al, 2020), preferring instead to turn to their peers for advice and support (Harvey et al. 2019, Tighe et al, 2017). The issue is further complicated in environments where harm reduction services are limited such as prisons or countries where such services are not available (Bates & Backhouse, 2020) and scant evidence of appropriate treatments (Bates et al, 2019a).
The evidence base for behavioural interventions with IPED users is also scant and is primarily based within a sport or anti-doping context (Bates et al, 2019b). Whilst there exists a small evidence base for behaviour change in this context, there are no very long-term evaluations of such programmes and no evidence of their applicability within the general population outside a sporting context.
There is a need to develop the research around effective and appropriate services for IPED users and it is hoped that this website will help facilitate global partnerships to address these issues.
References: Section F
Bates G, Van Hout MC, Teck JTW, McVeigh J. (2019a) Treatments for people who use anabolic androgenic steroids: a scoping review. Harm Reduction Journal.;16(1):75.
Bates G, Begley E, Tod D, Jones L, Leavey C, McVeigh J. (2019b) A systematic review investigating the behaviour change strategies in interventions to prevent misuse of anabolic steroids. J Health Psychol.;24(11):1595-612
Bates, G. & Backhouse, S. (2020) Preventing image and performance enhancing drug use: It’s not all chalk and talk. In K. Van de Ven, KJD Mulrooney, & J McVeigh, (Eds) Human Enhancement Drugs. (Ch. 20, pp280-297) Abingdon: Routledge
Harvey OA, Keem S, Parriosh M, van Teijlingen E. (2019) Support for people who use Anabolic Androgenic Steroids: A Systematic Scoping Review into what they want and what they access. BMC Public Health;19:1024.
Hope, V. Leavy, C., Morgan, G., Acreman, D., Turner, D. & Smith, J. (2020) Facilitators and barriers to health care access amongst people using image and performance enhancing drugs in Wales: Findings & Outcomes Report. Public Health Wales
McVeigh, J., Salinas, M. & Ralphs, R. (2020) A sentinel population: The public health benefits of monitoring enhanced body builders International Journal of Drug Policy https://doi.org/10.1016/j.drugpo.2020.102890
Tighe, B., M. Dunn, F. H. McKay & T. Piatkowski (2017). “Information sought, information shared: exploring performance and image enhancing drug user-facilitated harm reduction information in online forums.” Harm Reduction Journal 14(1): 48
Yu J, Hildebrandt T, Lanzieri N. (2015) Healthcare professionals’ stigmatization of men with anabolic androgenic steroid use and eating disorders. Body Image. Sep;15: 49-53. doi: 10.1016/j.bodyim.2015.06.001. Epub 2015 Jun 28. PMID: 26125091.
Zahnow, R., McVeigh, J., Ferris, J., & Winstock, A. (2017). Adverse Effects, Health Service Engagement, and Service Satisfaction Among Anabolic Androgenic Steroid Users. Contemporary Drug Problems, 44(1), 69-83. doi:10.1177/0091450917694268
